Colors Underwater
One of the first things you notice as a new scuba diver is the surprising lack of color. Discovery Channel specials and magazine layouts have brilliant colors. Where'd they all go?
Don't feel ripped off. The fact is, water absorbs light rapidly. So rapidly, that after only 300 ft (80 m), no visible light remains. This is far deeper than you'll ever go, but this absorption is very important at all depths.
The visible light spectrum can be broken up into the familiar
constituent colors (for example, see the too-familiar picture of Newton
with a prism). From least to highest energy, red, orange, yellow,
green, blue, and violet. This order is important, because it is the
lowest energy colors that are absorbed first in water. This chart below
shows the depths at which different colors are absorbed. These are
approximate, as water clarity and turbidity affect color absorption.

What is doing the absorbing exactly? All water contains microscopic particles. Light strikes these particles and scatters, with some of the light absorbed. What remains is what color you see. As the light travels farther, only blue light remains, with it eventually being absorbed as well^1^. Of course, flashlights reintroduce "white" light, which contains the entire spectrum, making all colors visible again.
To illustrate, I've recreated how some common items would look at
different depths.

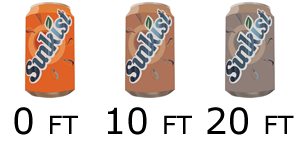
Notice how the red is removed from the orange, leaving a little color
while the blue text is unaffected.
1. Different particles scatter light differently, so some water appears more green than blue.
